CMS Will Pay for COVID-19 Booster Shots, Eligible Consumers Can Receive at No Cost
From CMS on September 24, 2021
CMS Will Pay for COVID-19 Booster Shots, Eligible Consumers Can Receive at No Cost
Coverage without cost-sharing available for eligible people with Medicare, Medicaid, CHIP, and Most Commercial Health Insurance Coverage
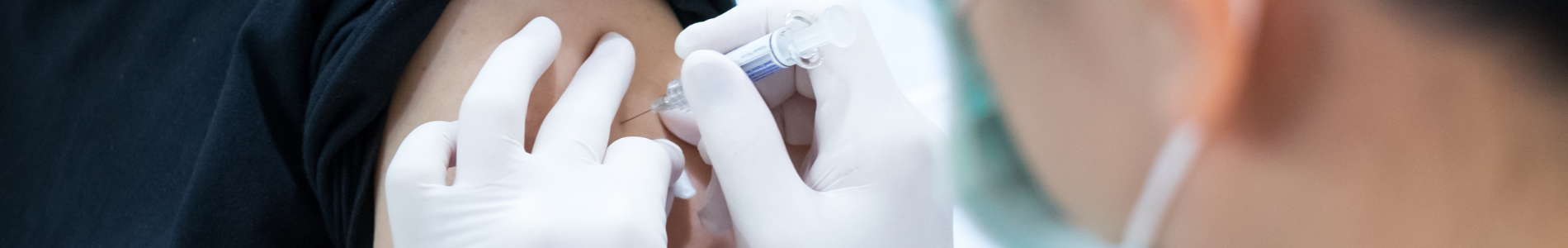
Following the Food and Drug Administration’s (FDA) recent action that authorized a booster dose of the Pfizer COVID-19 vaccine for certain high-risk populations and a recommendation from the Centers for Disease Control and Prevention (CDC), the Centers for Medicare & Medicaid Services (CMS) will continue to provide coverage for this critical protection from the virus, including booster doses, without cost sharing.
Beneficiaries with Medicare pay nothing for COVID-19 vaccines or their administration, and there is no applicable copayment, coinsurance or deductible. In addition, thanks to the American Rescue Plan Act of 2021 (ARP), nearly all Medicaid and CHIP beneficiaries must receive coverage of COVID-19 vaccines and their administration, without cost-sharing. COVID-19 vaccines and their administration, including boosters, will also be covered without cost-sharing for eligible consumers of most issuers of health insurance in the commercial market. People can visit vaccines.gov (English) or vacunas.gov (Spanish) to search for vaccines nearby.
“The Biden-Harris Administration has made the safe and effective COVID-19 vaccines accessible and free to people across the country. CMS is ensuring that cost is not a barrier to access, including for boosters,” said CMS Administrator Chiquita Brooks-LaSure. “CMS will pay Medicare vaccine providers who administer approved COVID-19 boosters, enabling people to access these vaccines at no cost.”
CMS continues to explore ways to ensure maximum access to COVID-19 vaccinations. More information regarding the CDC COVID-19 Vaccination Program Provider Requirements and how the COVID-19 vaccine is provided through that program at no cost to recipients is available at https://www.cdc.gov/
###
Get CMS news at cms.gov/newsroom, sign up for CMS news via email and follow CMS on Twitter @CMSgov